 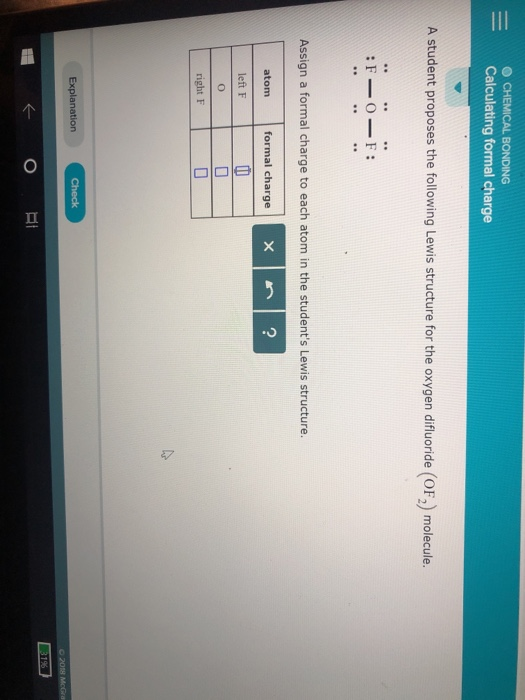
This is also referred to as the “centre” position. Typically, the less electronegative atom occupies the central position in the molecule. The formal charge of an individual atom in a Lewis structure cannot be zero, as each atom has a unique charge. When calculating the formal charge of an individual atom, images are often used to illustrate electrons and bonds between atoms. The formal charge is a measure of the electrons that each atom has and determines whether the Lewis structure of the molecule is correct. 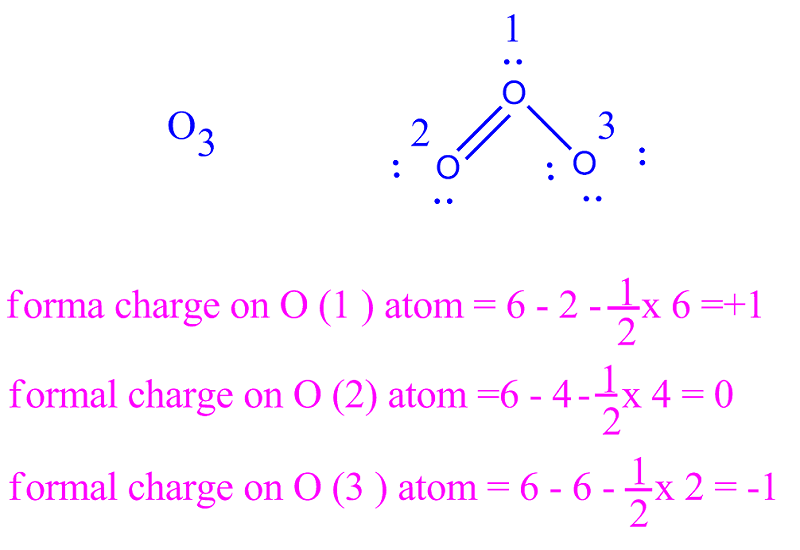
The formal charge of an atom is a very important concept in chemistry, since it is required to understand ionic charge and molecular structures. 
All atoms have a zero charge, and this is the reason why the formal charge of an individual atom is different than the actual one. The difference between these values is called the formal charge. When calculating formal charge, the lone pair and bonding electrons are counted separately. Atoms are assigned this charge based on the number of electrons they carry in their bonds and the number of valence electrons in a single atom. The formal charge of an individual atom is the amount of energy an atomic carries in its electronic state. Formal charge is the electrical charge of an individual atom If you have questions about formal charge, feel free to ask them in the comments below. The information provided should be useful for all levels of chemical students. This article will explain how to calculate the formal charge and what it means. We can use this information to calculate the preferred Lewis structure of a molecule. The formal charge is derived from the number of valence electrons in a neutral atom. In chemistry, we use the theoretical charge of individual atoms to predict the Lewis structure of molecules. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
